Difference between revisions of "QIBA Profile Conformance"
| Line 42: | Line 42: | ||
:'''Checklist for sites''' | :'''Checklist for sites''' | ||
| + | * [[Media:QIBA FDG-PET/CT Conformance Checklist 2021-03-31.pdf | QIBA FDG-PET/CT Conformance Checklist 2021-03-31]] | ||
:'''Checklist for clinical trial''' | :'''Checklist for clinical trial''' | ||
Revision as of 21:14, 13 April 2021
Self-attestation (Registered) or QIBA Tested (Certified) Opportunities
{This content will eventually be available on the RSNA.org website.}
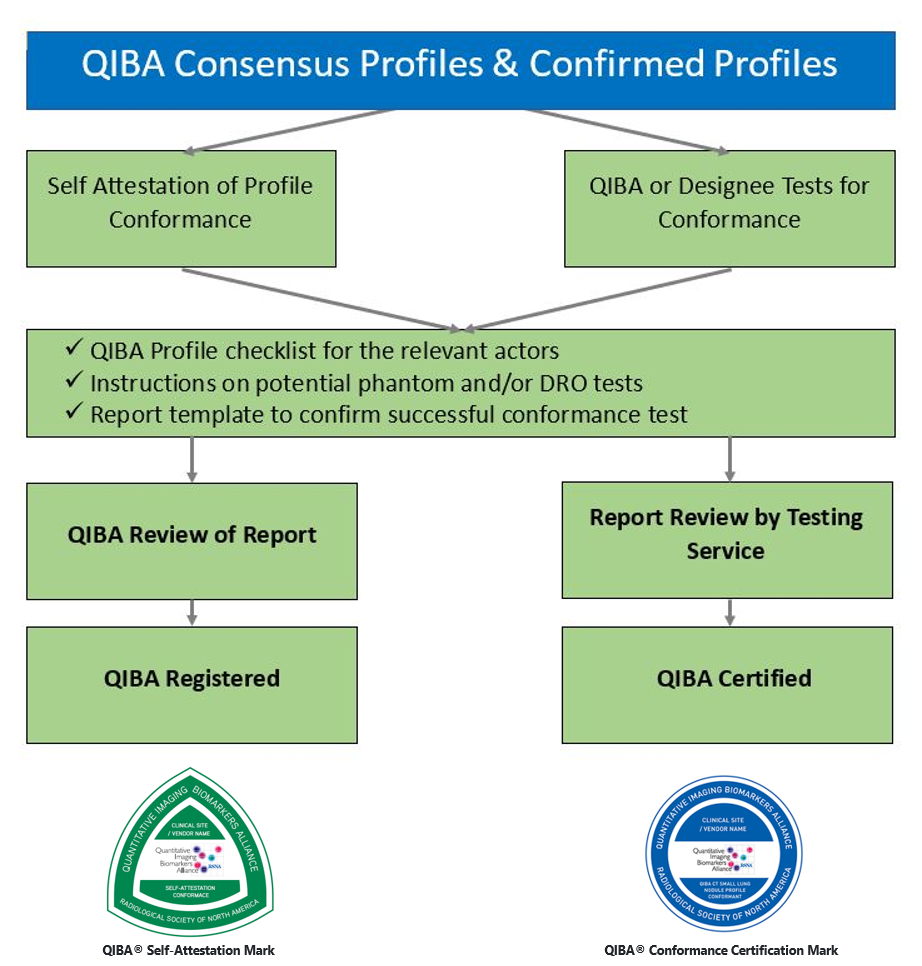
QIBA Profiles that are at least at the Consensus Stage are available to interested parties (clinical sites, CROs, equipment and/or software vendors) to demonstrate that all relevant actors of an institution or product conform to the respective QIBA Profile.
As shown in the following diagram, QIBA Profile conformance may be demonstrated via Self Attestation, or (when available) via a Certification service:
Before starting conformance testing, please Email QIBA
Certification Service for Conformance
CT Small Lung Nodule Volumetry for Screening
- CT Small Lung Nodule Volume Assessment and Monitoring in Low Dose Screening 2018-11-18
- https://www.rsna.org/en/research/quantitative-imaging-biomarkers-alliance/qiba-conformance-certification-services
Self Attestation of Conformance
The following QIBA Profiles have reached the Consensus Stage or the Technically Confirmed Stage, and are thus appropriate for self-attestation conformance testing.
CT: Lung Tumor Volume (Advanced Disease)
- Profile
- Checklist
- Conformance Checklist (contains Appendix C extracted from the Profile document - pg 42)
- Assessment Procedures (contains Section 4 extracted from the Profile document - pg 30)
- Report Template
FDG-PET: Solid Tumor SUV
- Profile
- Checklist
- Checklist for sites
- Checklist for clinical trial
- Reporting
Please fill out your respective checklist and Email QIBA for conformance assessment.
DWI MR: ADC of Lesions in Four Organs
- Profile
- Checklist for sites
- Checklist for software providers
- Checklist for clinical trial
- Reporting
Please fill out your respective checklist and Email QIBA for conformance assessment.